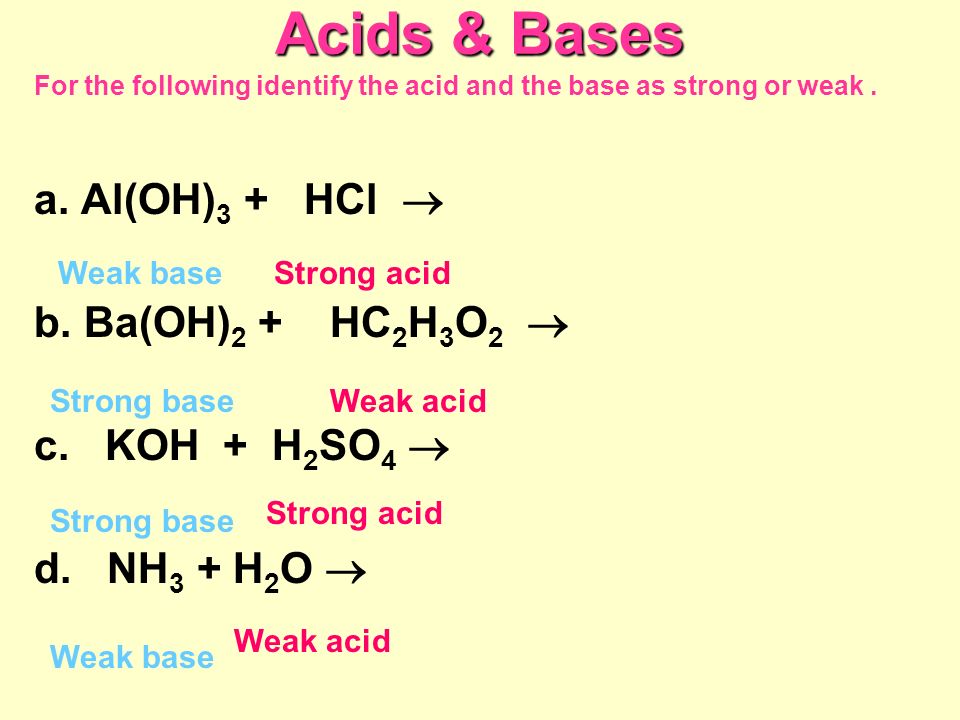
A. Calculate the pH of a 0.10 M solution of barium hydroxide, Ba(OH)2 - Home Work Help - Learn CBSE Forum

SOLVED: A solution of Ba(OH)2 was standardized against 0.1215 g of benzoic acid with grade of primary standard, C6H5COOH (122.12 g / mol). The end point was observed after adding 43.25 mL

A. Calculate the pH of a 0.10 M solution of barium hydroxide, Ba(OH)2 - Home Work Help - Learn CBSE Forum

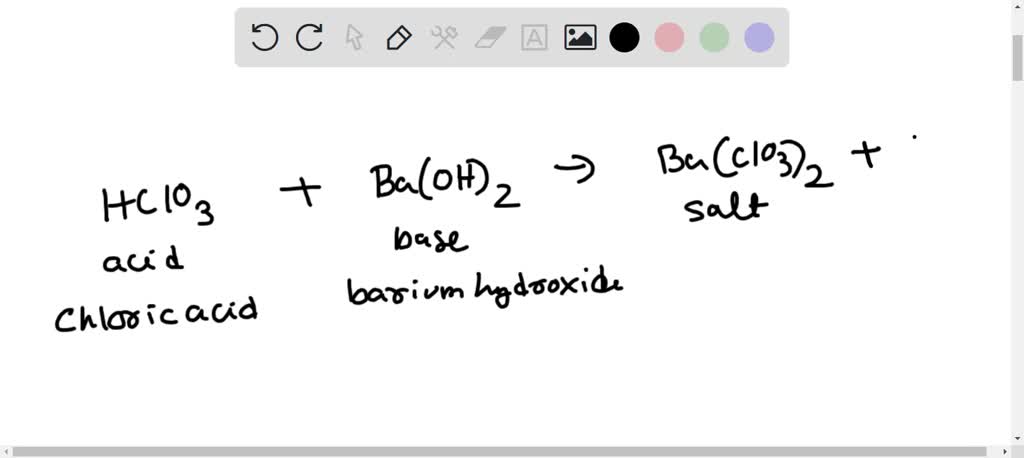
SOLVED: Write the balanced molecular equation for the neutralization reaction between HI and Ba(OH)2 in aqueous solution. Include physical states.

Question Video: Determining the Products of the Neutralization Reaction of Barium Hydroxide Ba(OH)₂ with Carbonic Acid H₂CO₃ | Nagwa

Calculate the pH of a 0.10 M solution of barium hydroxide, Ba(OH)2. Express your answer numerically - Home Work Help - Learn CBSE Forum