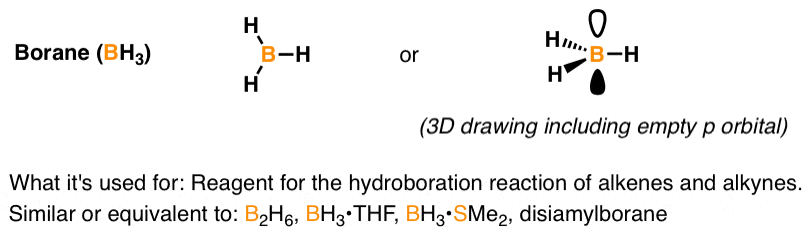
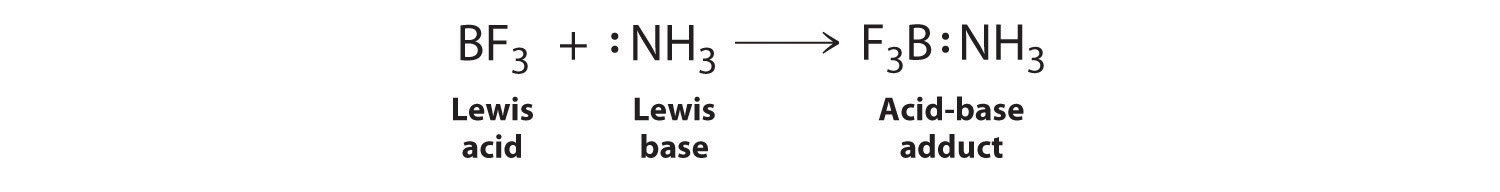
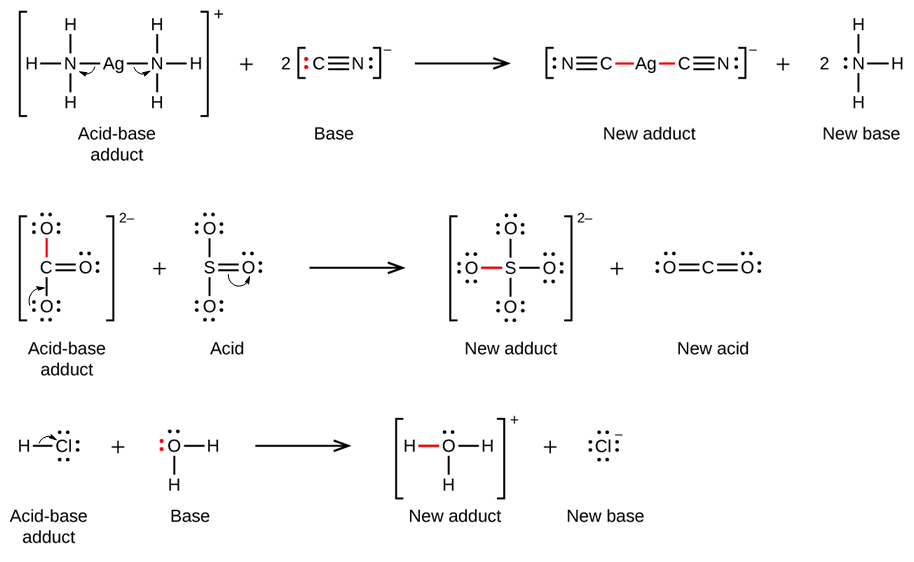
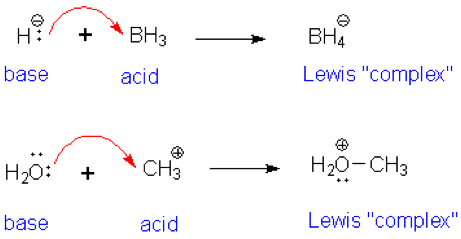
When NH3 reacts with BH3, a boron-nitrogen bond is formed joining the two compounds. Which is the electrophile in this reaction? a. Electrophile BH3 b. Nucleophile NH3 | Homework.Study.com
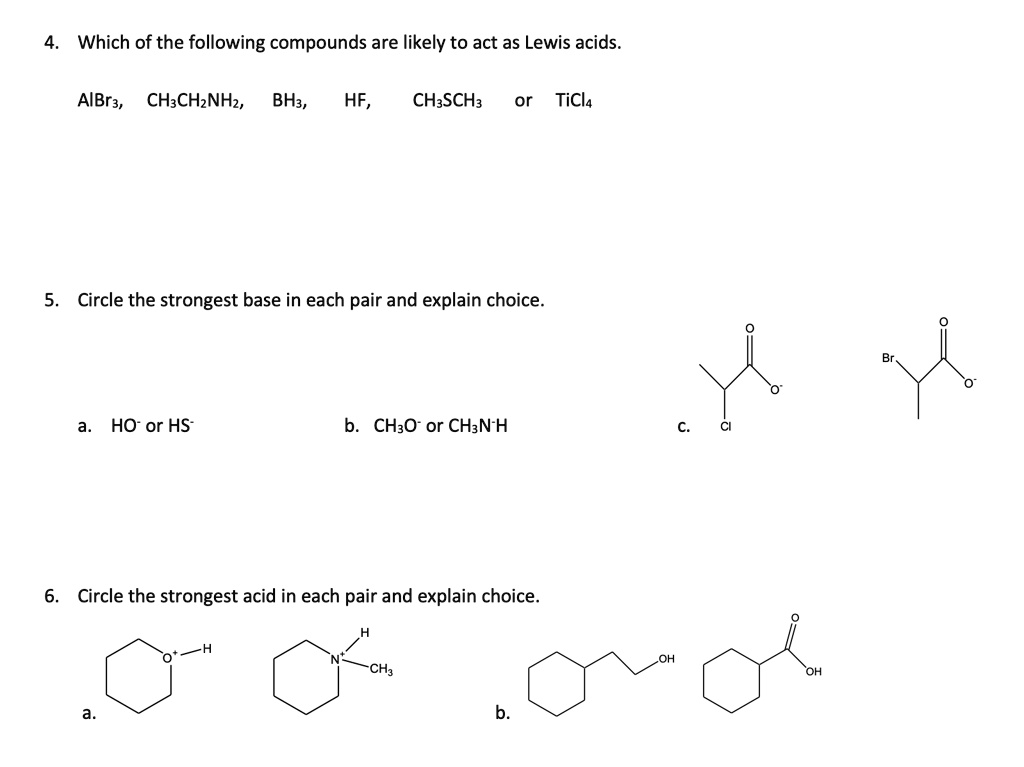
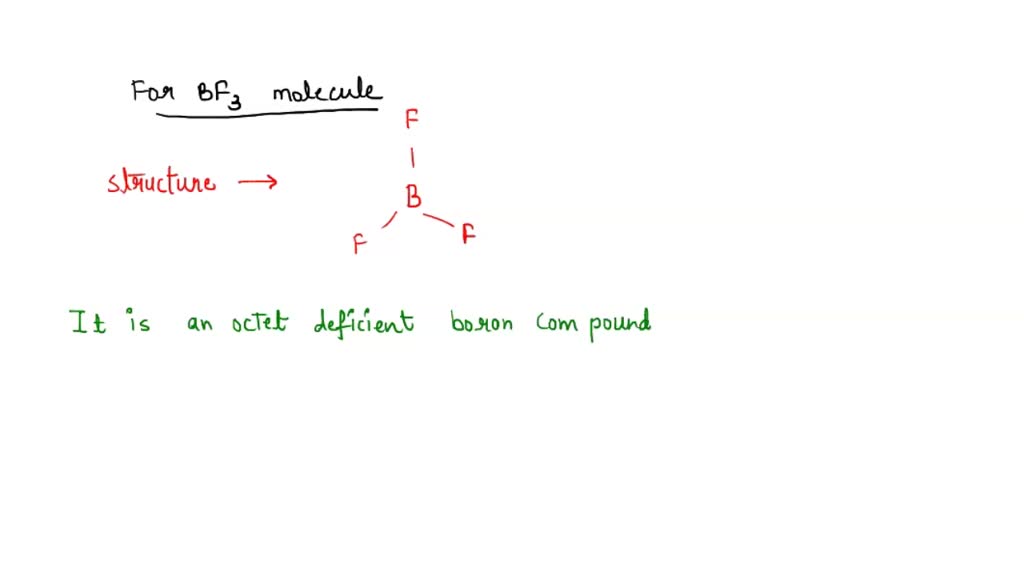
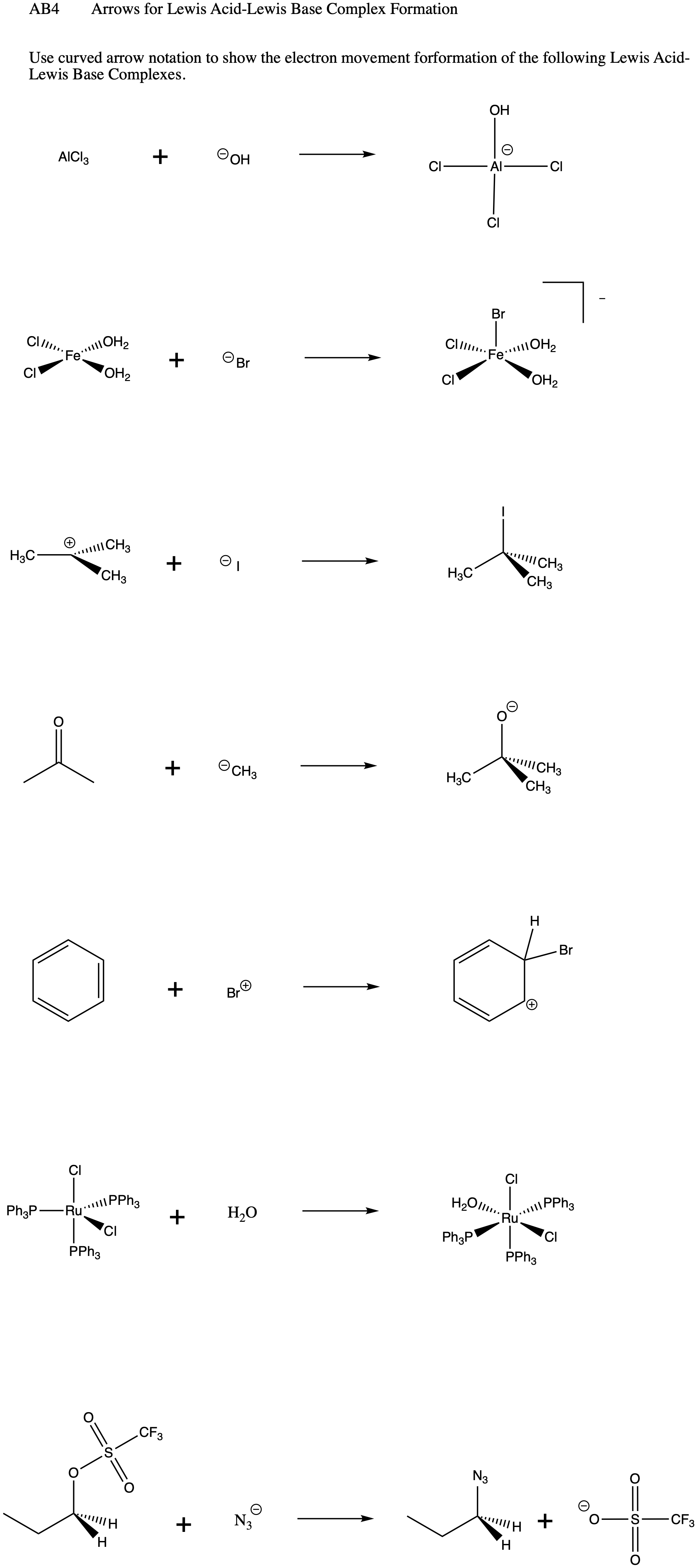
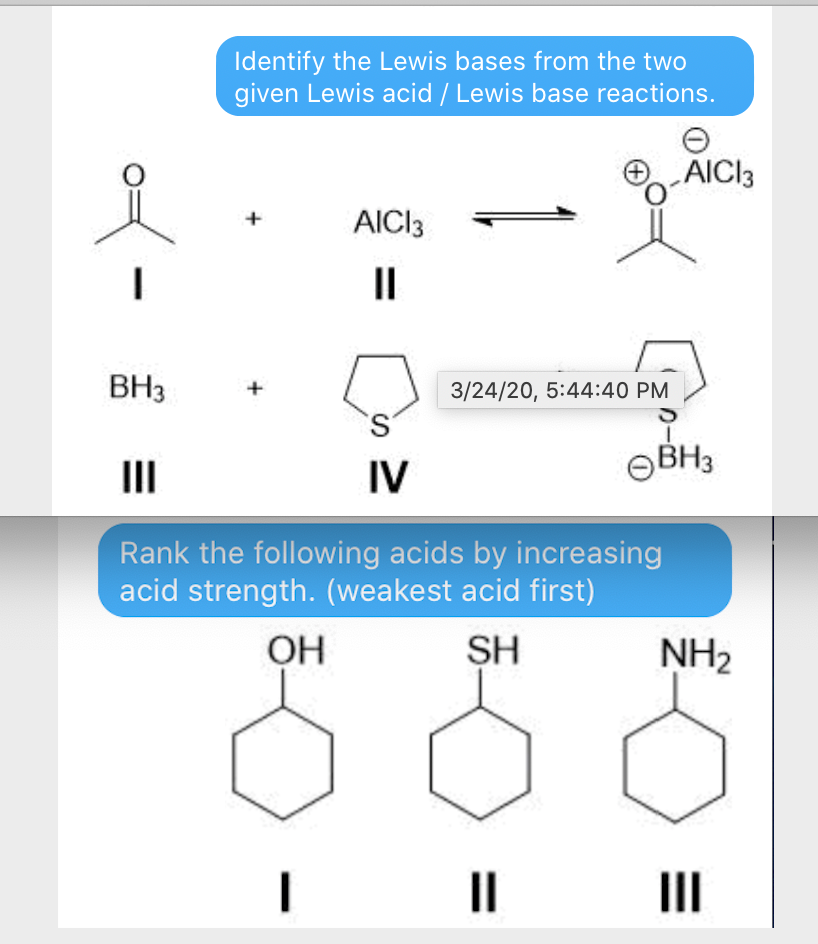
SOLVED: Which of the following compounds are likely to act as Lewis acids. AIBr3, CH3CHzNHz, BH3, HF, CH;SCH3 TiCla Circle the strongest base in each pair and explain choice HO or HS
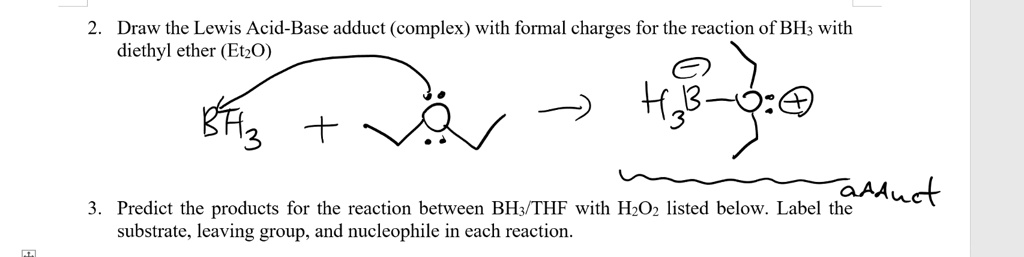
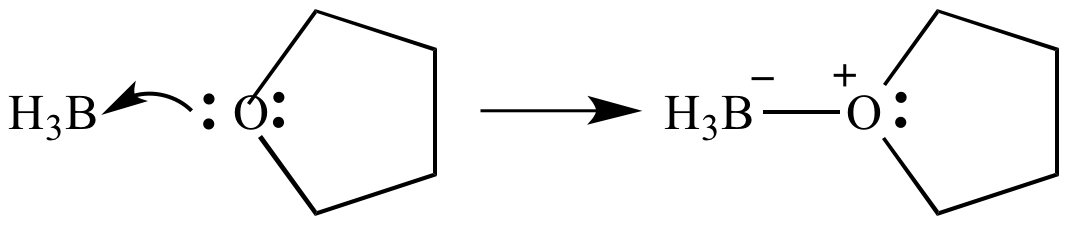
SOLVED: Draw the Lewis Acid-Base adduct (complex) with formal charges for the reaction of BH; with diethyl ether (EtzO) aAAuct Predict the products for the reaction between BH3 THF with HzOz listed
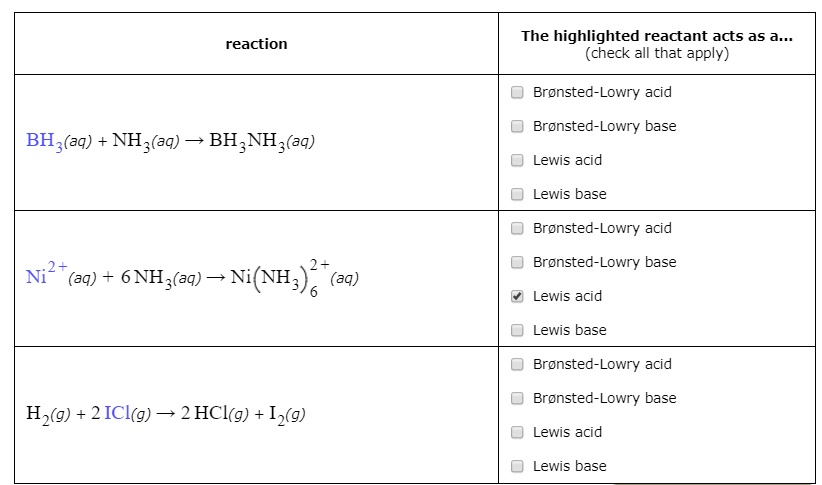
SOLVED: The highlighted reactant acts as a (check all that apply) reaction Bronsted-Lowry acid Bronsted-Lowry base BHz(aq) NHz(aq) BH3NH3(aq) Lewis acid Lewis base Bronsted-Lowry acid Bronsted-Lowry base Ni (aq) 6 NH3(aq) Ni(NH;) (