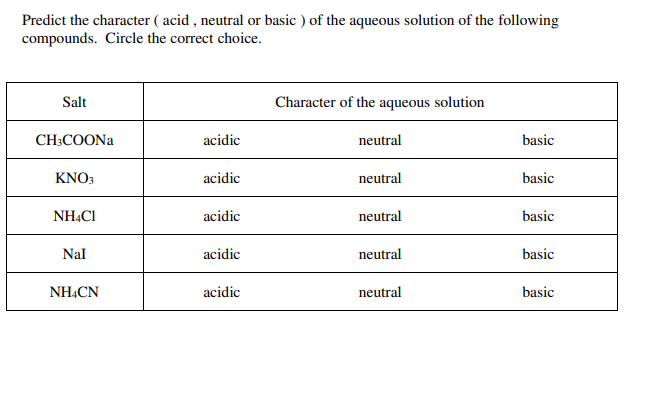
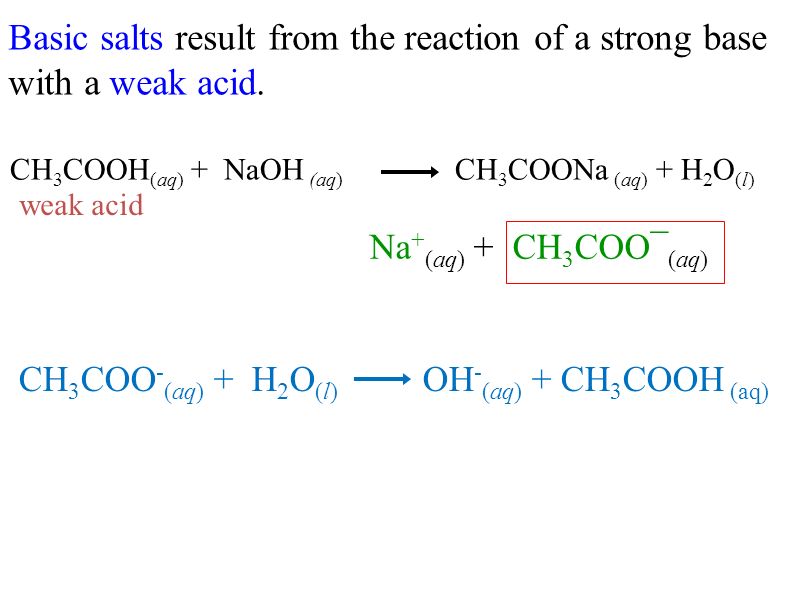
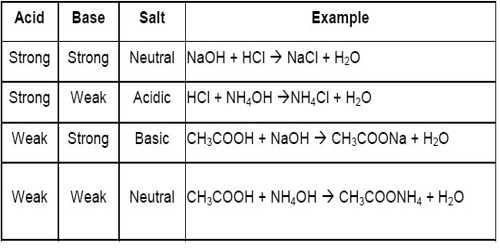
classify the following as acidic basic or neutral salt NaCl, classify the following as acidic basic or - Brainly.in

SOLVED: A buffer is prepared using acetic acid, CH3COOH, (a weak acid, pKa = 4.75) and sodium acetate, CH3COONa (which provides acetate ions, the conjugate base), according to the following proportions: Volume

You have 250mL of a 0.56M solution of sodium acetate. How many mL of 0.50M acetic acid should be added to make a buffer of pH 4.40? | Homework.Study.com

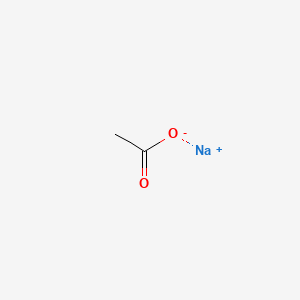
Sodium Acetate(CH3COONa) - Structure, Properties, Preparations, Uses, Important questions, FAQs of sodium acetate.
![SOLVED: Theme: Acid-base balance Determine the pH of a solution of sodium acetate [CH3COONa] which is 0.150 M. Then, determine the % hydrolysis SOLVED: Theme: Acid-base balance Determine the pH of a solution of sodium acetate [CH3COONa] which is 0.150 M. Then, determine the % hydrolysis](https://cdn.numerade.com/ask_previews/567a4379-b31b-4987-9817-e40d2cf36c2a_large.jpg)
SOLVED: Theme: Acid-base balance Determine the pH of a solution of sodium acetate [CH3COONa] which is 0.150 M. Then, determine the % hydrolysis