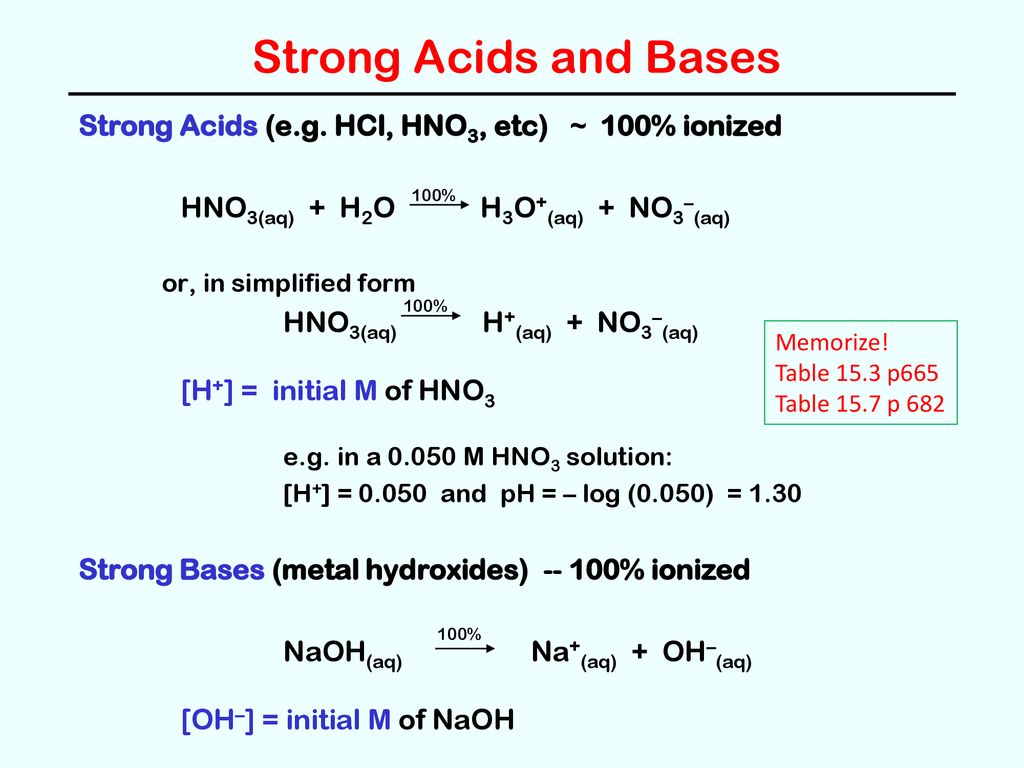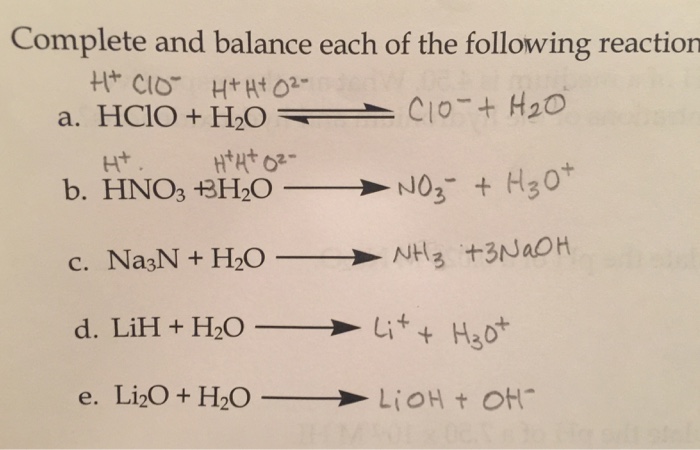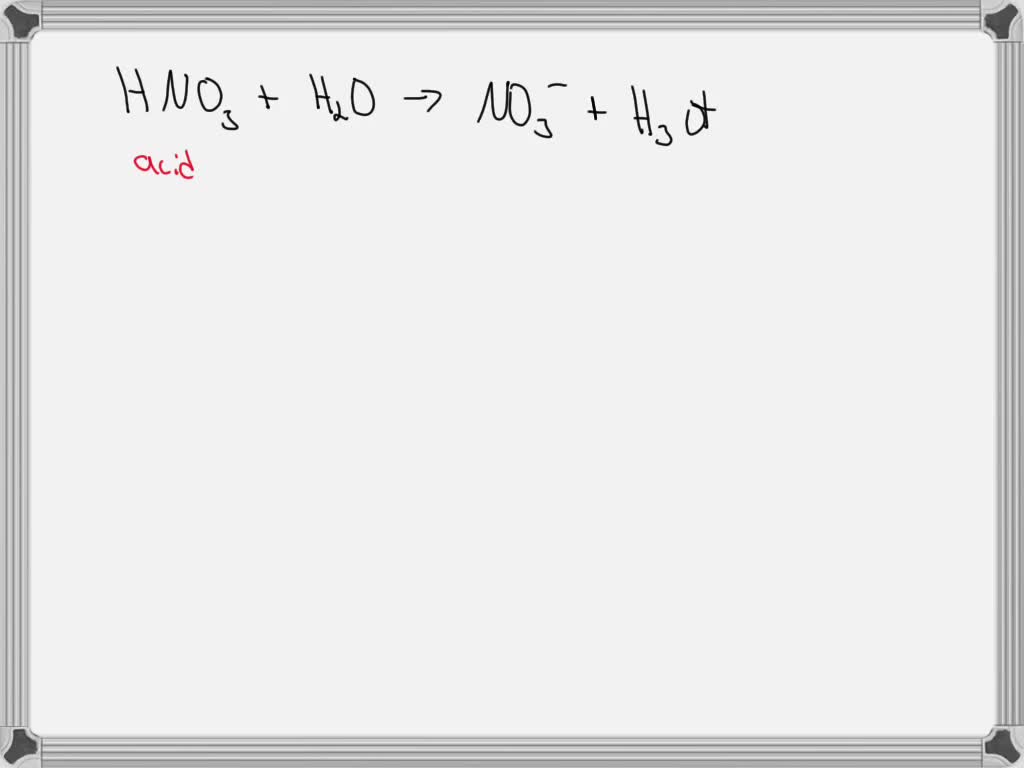
SOLVED: Identify the conjugate acid in the following reaction, HNO3(aq) + H2O(l) → NO3- (aq) + H3O+ (aq). H3O+ NO3- HNO3 H2O None of the above
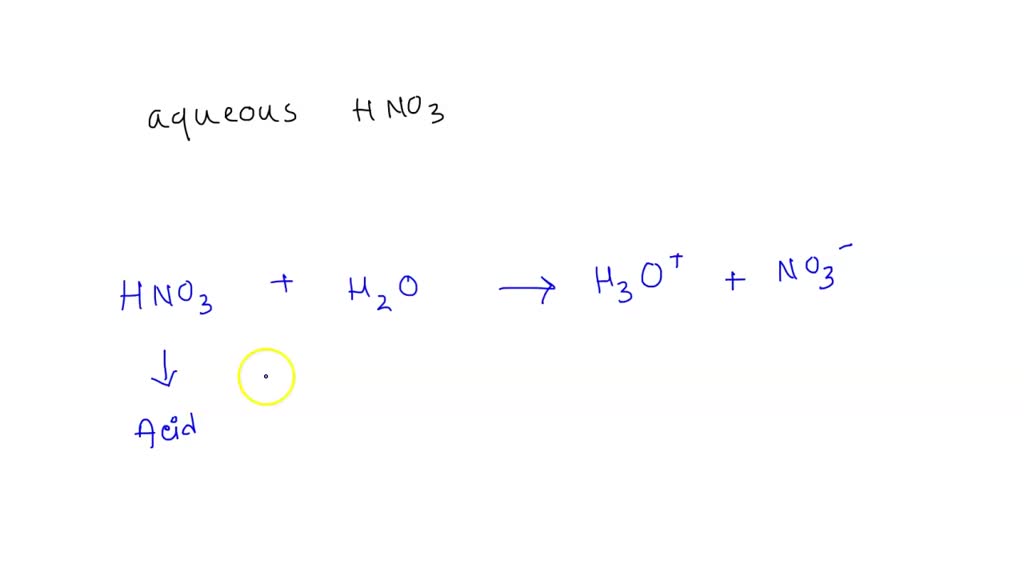
SOLVED: Which of the following would be the correct products for the following acid /base equation? HNO3 is the acid. HNO3 + H2O –> ? acid A.)H2NO3+ + H3O+ B.)H2NO3+ + OH-

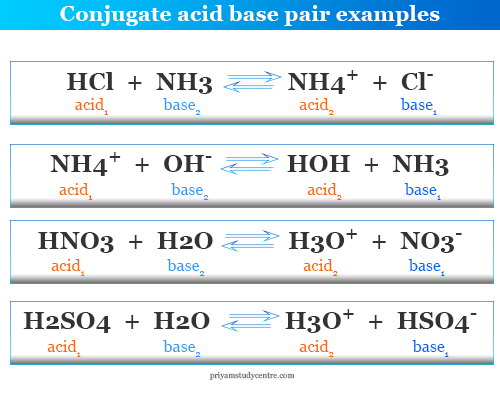
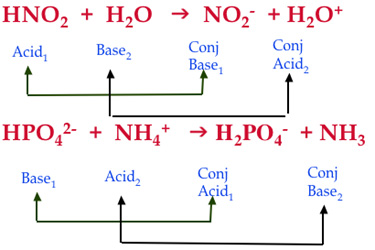
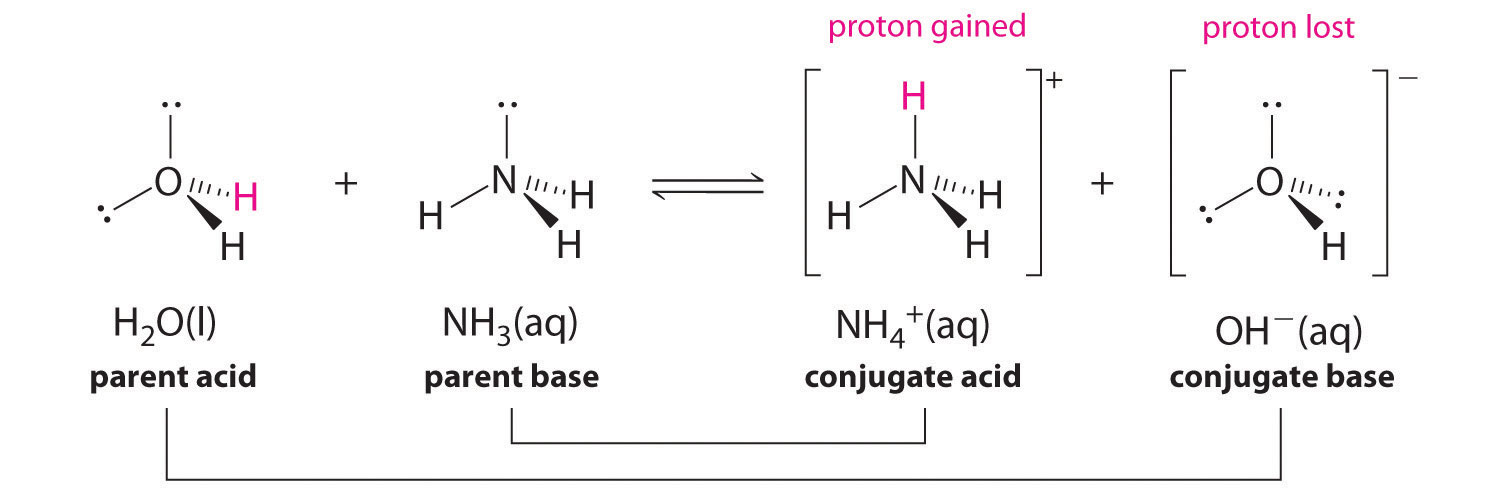
Identify the acid, base, conjugate acid, and conjugate base in the following reaction. HNO3 + NH3 + NO3 + - Brainly.com

Question Video: Calculating the Concentration of Nitric Acid via Titrating against a Known Volume of Potassium Hydroxide | Nagwa

Nucleation of Mixed Nitric Acid–Water Ice Nanoparticles in Molecular Beams that Starts with a HNO3 Molecule | The Journal of Physical Chemistry Letters