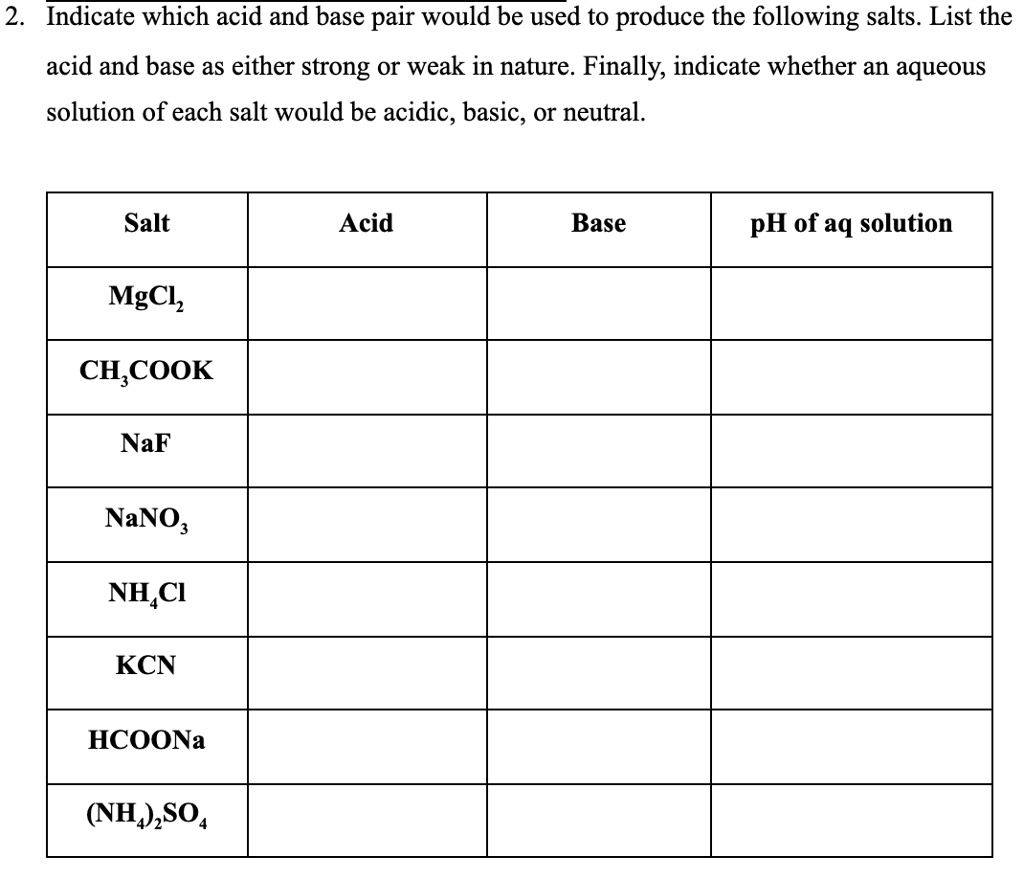
How to Determine if Salt is Acidic, Basic, or Neutral Example, Problem, Shortcut, Explained Question - YouTube

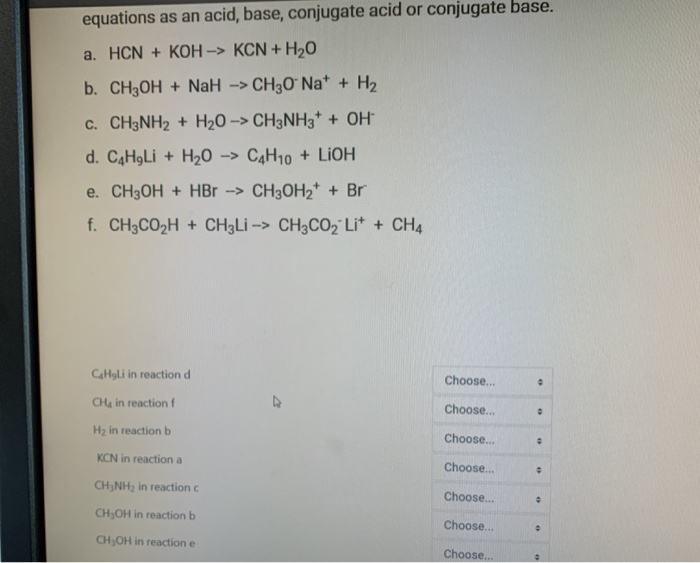
OneClass: Consider the following data on some weak acids and weak bases: acid base C0 name formula na...

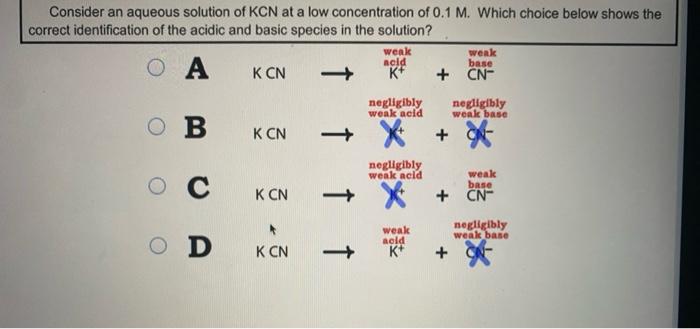
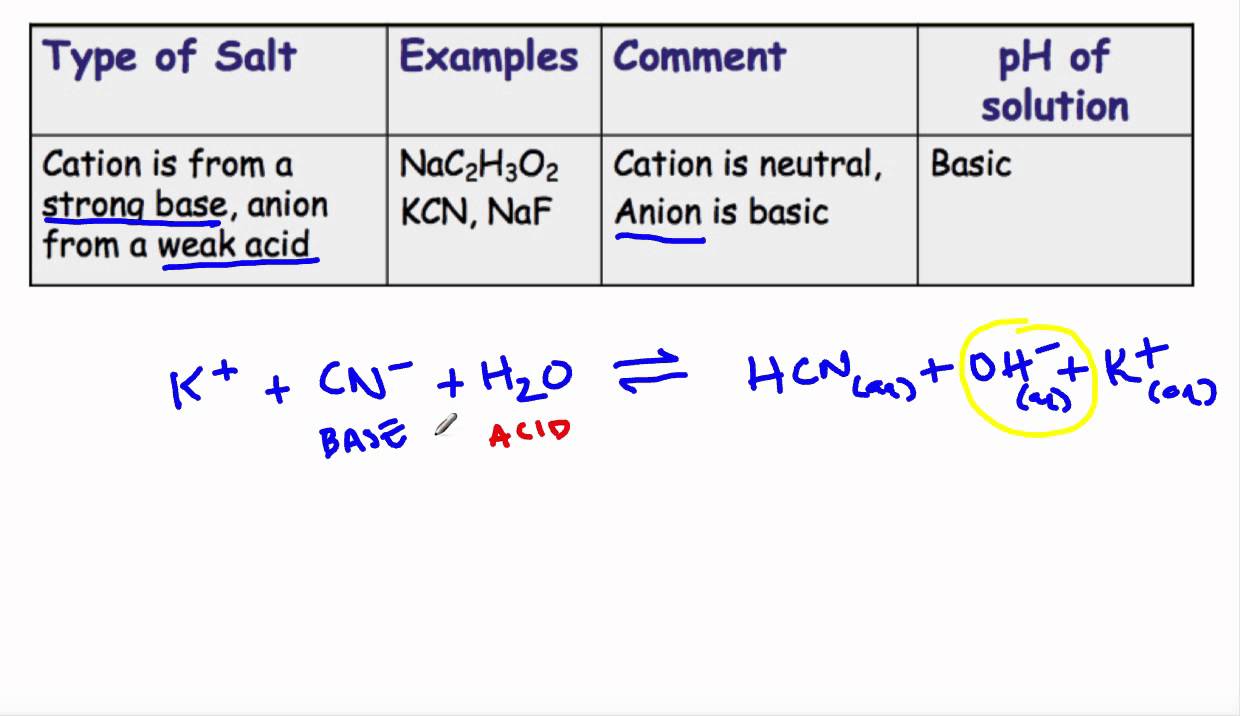
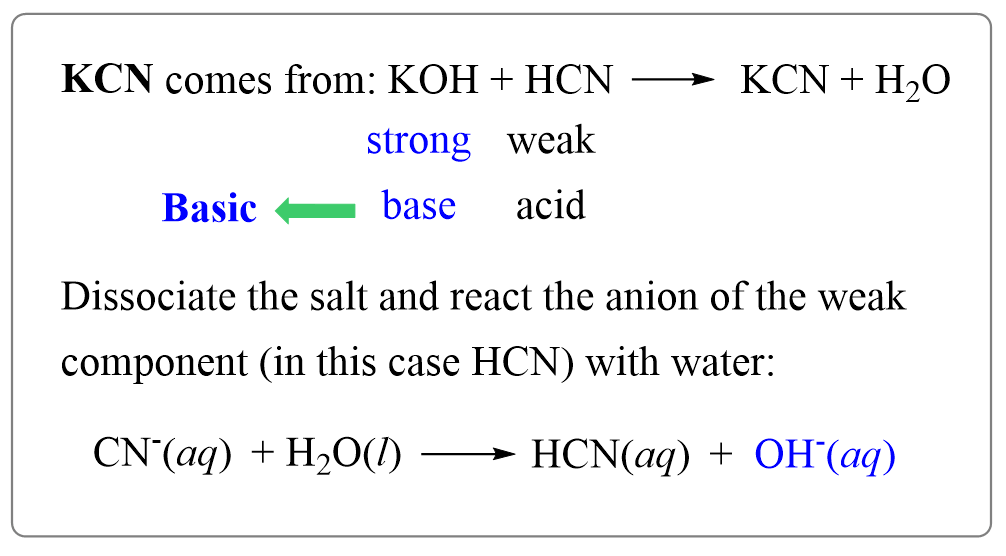
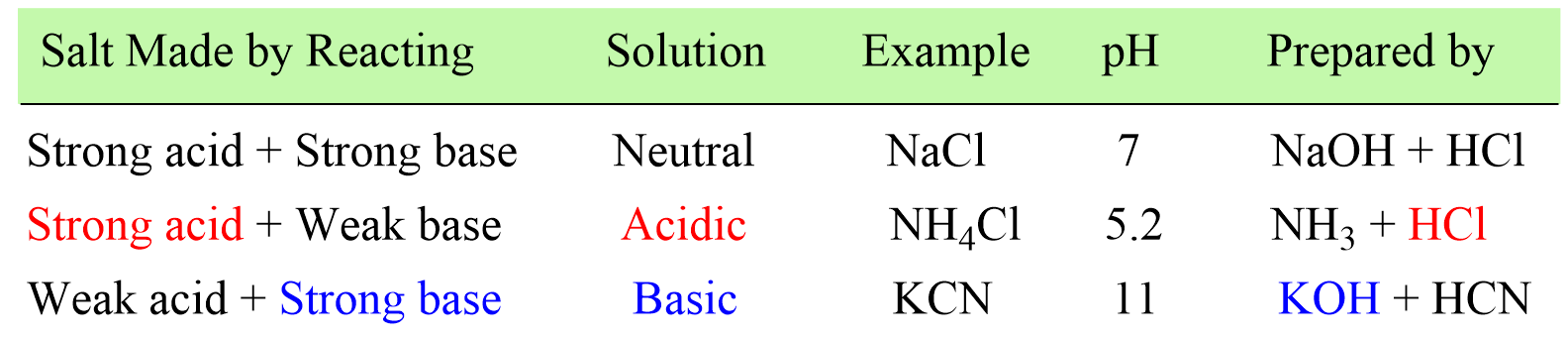
When potassium cyanide reacts with water, will the resulting solution be acidic, alkaline or neutral? Justify your answer.

When potassium cyanide reacts with water, will the resulting solution be acidic, alkaline or neutral? Justify your answer.

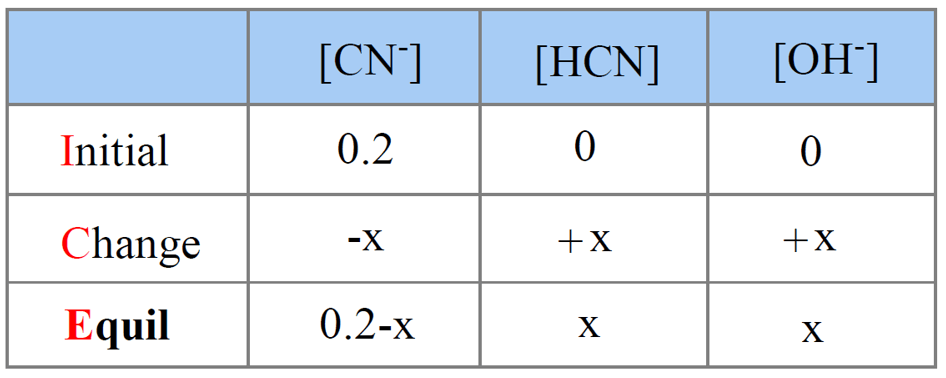
SOLVED: Example: Calculate the pH ofa 0.500 M solution of KCN For HCN, K = 5.8 x 10 -10 For CN , then, Kb (SHOW WORK) R 0.500 c E X 0.500 -x 0 X +X +X