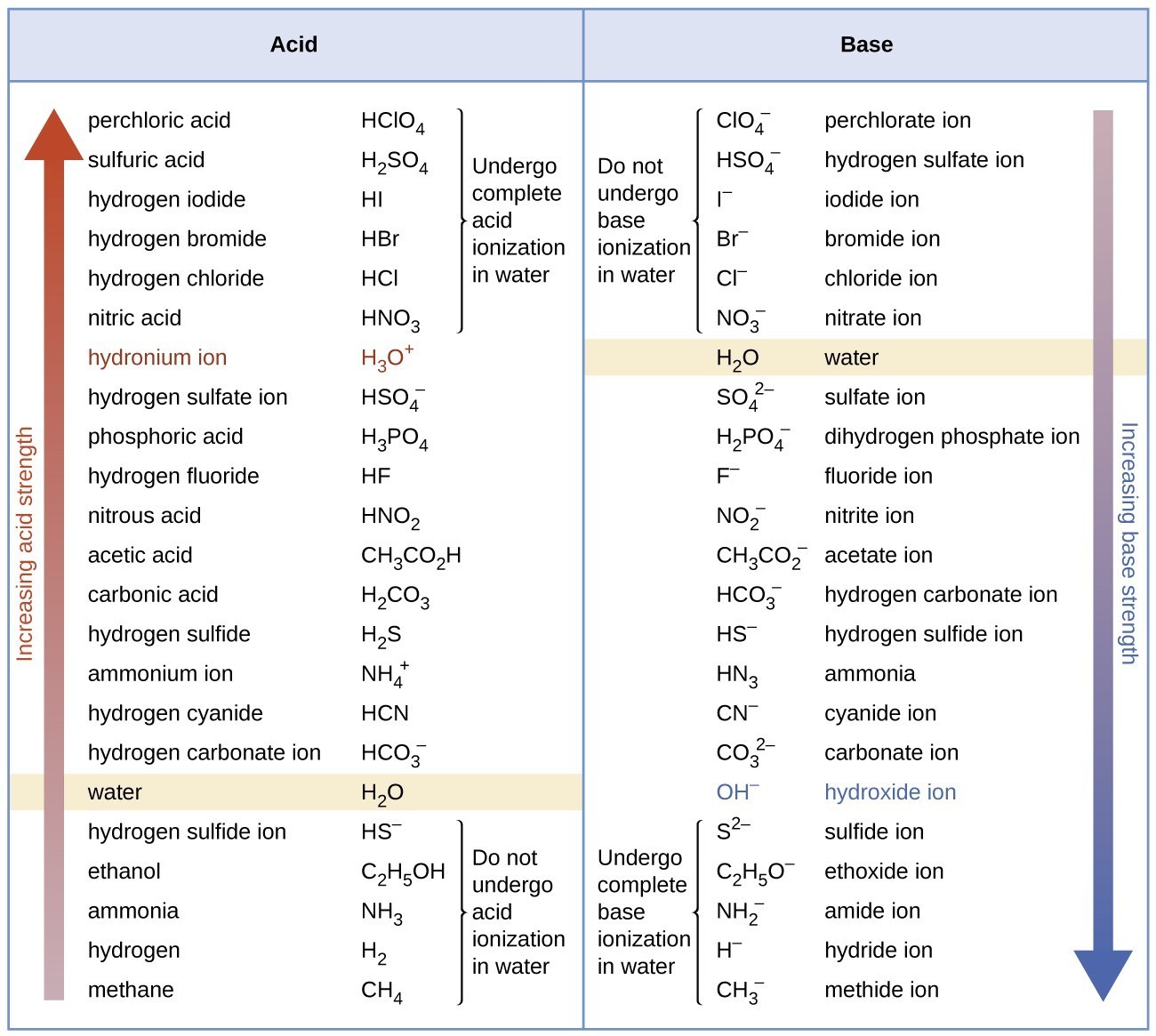
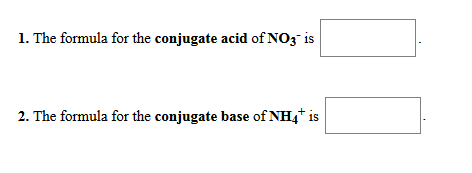Morphine (C(17)H(19)NO(3)), Which is used medically to relieve to pain is a base. What is its conjugate acid?

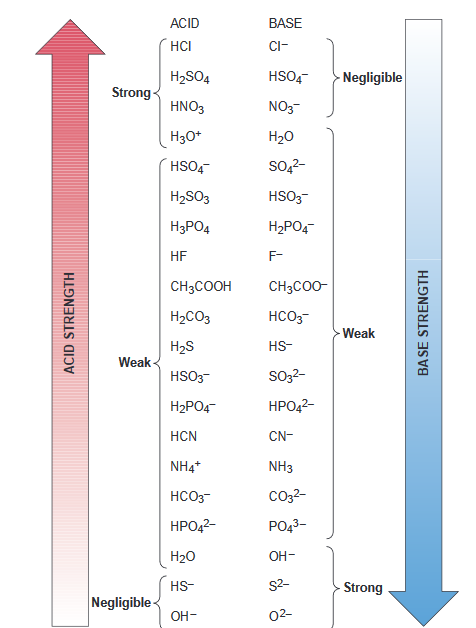
Fillable Online UNIT 14 - Acids & Bases ACID BASE HSO4 H3PO4 NO3 H2PO4 ... Fax Email Print - pdfFiller

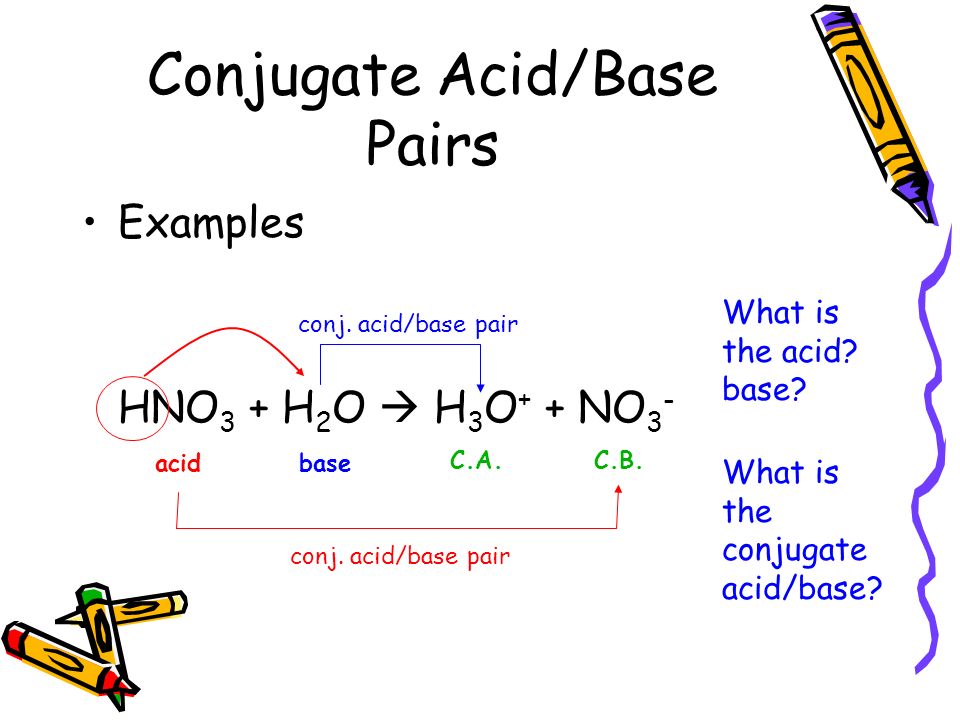
⚗️Identify the acid, base, conjugate acid, and conjugate base in the following reaction. HNO3 + NH3 + - Brainly.com
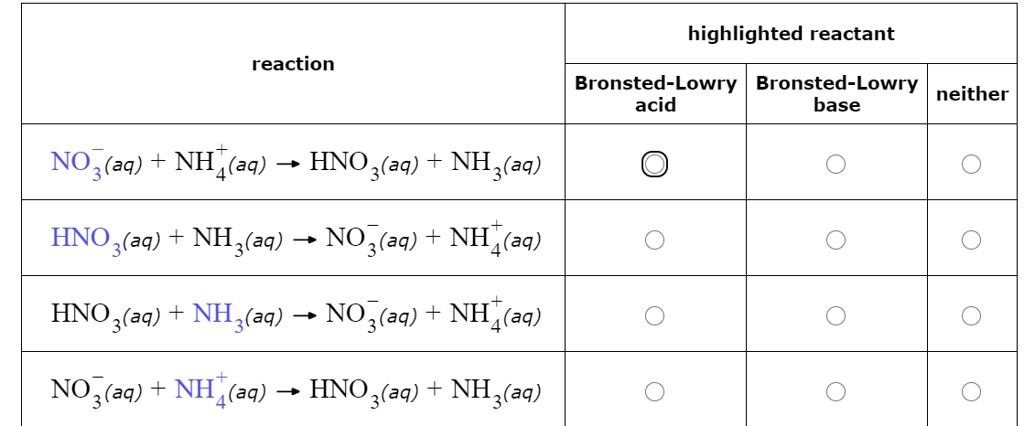
SOLVED: highlighted reactant reaction Bronsted-Lowry Bronsted-Lowry neither acid base NO3(aq) + NH4(aq) HNO 3(aq) NH,(aq) HNO 3(aq) + NH3(aq) 3 NO3(aq) NH A (aq) HNO 3(aq) + NH3(aq) 3 NO3(aq) NH A (
Acids And Bases. Section 19.1 Acid-Base Theories OBJECTIVES: –Define the properties of acids and bases. - ppt download

Classify each of these reactions. redox acid -base neutralization precipitation Pb (NO3) 2(aq) + 2 KCI(aq) - Home Work Help - Learn CBSE Forum

OneClass: 26 Which set is a conjugate acid/base pair? Marks: 1 Choose one answer a. HNO3 NO2 b. HNO2 ...
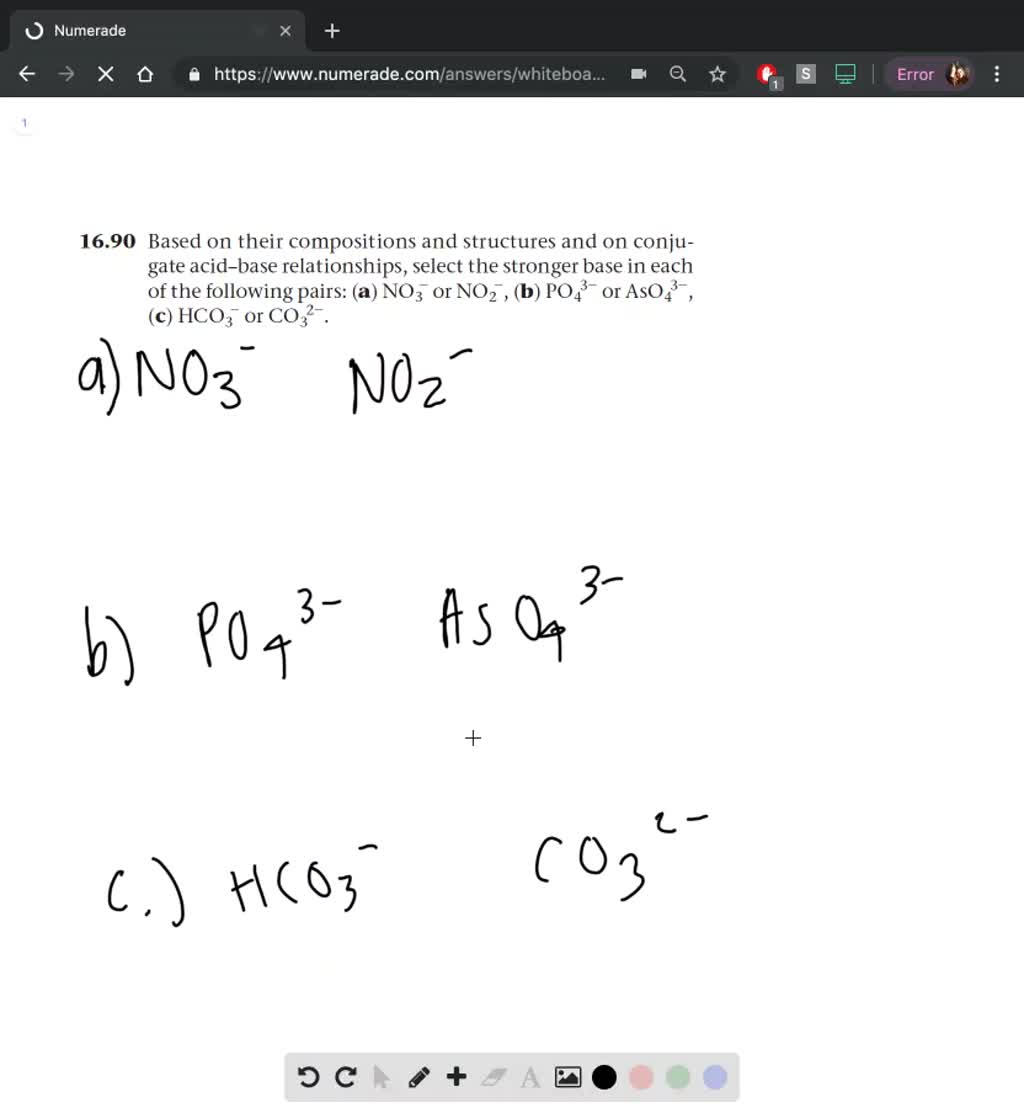
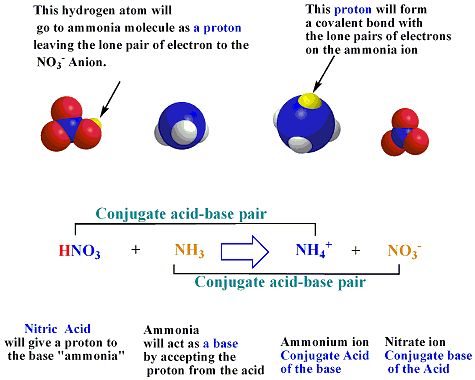


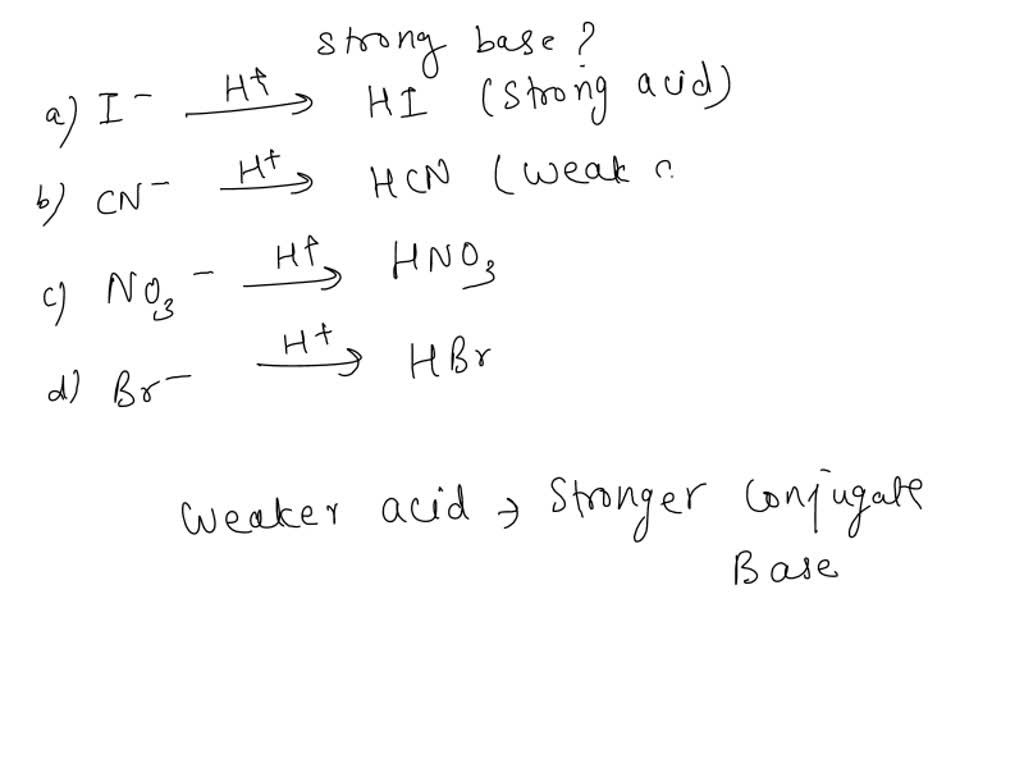


![Mixing curve of low base flow [NO3 -] with shallow flow [NO3 -]... | Download Scientific Diagram Mixing curve of low base flow [NO3 -] with shallow flow [NO3 -]... | Download Scientific Diagram](https://www.researchgate.net/publication/346661459/figure/fig4/AS:965884747198464@1607296277040/Mixing-curve-of-low-base-flow-NO3-with-shallow-flow-NO3-superposed-onto-weekly.png)